Metals in group 3 – metals in group 3 need to lose three electrons, so become ions with a +3 charge.For example, beryllium (Be) loses two electrons to become Be 2+, magnesium (Mg) loses two electrons to become Mg 2+ and calcium (Ca) loses two electrons to become Ca 2+. Metals in group 2 – all metals in group 2 need to lose two electrons, so become ions with a +2 charge.For example, lithium (Li) loses one electron to become Li +, sodium (Na) loses one electron to become Na + and potassium (K) loses one electron to become K +. Metals in group 1 – all metals in group 1 need to lose one electron, so become ions with a +1 charge.When an atom of magnesium loses two electrons, it becomes a positively charged magnesium ion represented as M g 2+įor your exam, you must know the charges of the ions formed from the metals in groups 1, 2 and 3 and the non-metals in groups 5, 6 and 7: For example, an oxygen atom could gain one electron to become an oxide ion with a single negative charge which would be represented as, or it could gain two electrons to become an oxide ion represented as. Charges on ions are always shown as superscript numbers given after the chemical symbol for the atom or molecule. The overall charge on an ion shows the number of electrons that have been gained or lost to form the ion. To achieve a full outer shell, the fluorine atom gains one electron to become a fluoride ion represented as F –. This means that they will always gain electrons to become anions.įor example, fluorine has seven electrons in its outer shell. Non-metal elements found on the right-hand side of the periodic table, all have outer electron shells which are almost full. By losing this electron the lithium atom becomes a lithium ion, which is represented as Li +. For example, lithium has one electron in its outer shell which it needs to lose. Metal elements in groups 1, 2 and 3 of the periodic table all lose electrons to become metal cations. The loss of electrons forms positive ions known as cations and the gain of electrons forms negative ions known as anions. Elements which only lose electrons will only become positive ions and those which can only gain electrons will only become negative ions. The number of electrons gained or lost determines the charge on the ion. This means that some elements will become ions only by losing electrons and some will become ions only by gaining electrons. In this case it is much easier for the atom to gain one electron that lose the seven from its outer shell. If an atom had seven electrons in its outer shell, it would need to either gain one electron or lose seven. It takes far less energy and is therefore much easier for the atom to lose one electron than gain seven. The number of electrons in an element’s outer shell determines the number of electrons it will gain or lose to achieve a full outer shell.įor example, an atom with one electron in its outer shell, will need to either gain seven electrons or lose one to gain the stability of a full outer shell. The group number of an element tells you the number of electrons it has in its outer shell.Īll atoms will react to achieve a full outer shell of electrons. For example, the ionic compound sodium chloride is formed when a positively charged sodium ion is attracted to a negatively charged chloride ion.Įlements in the periodic table are arranged in groups and periods. The force of attraction between oppositely charged ions causes them to form an ionic compound. 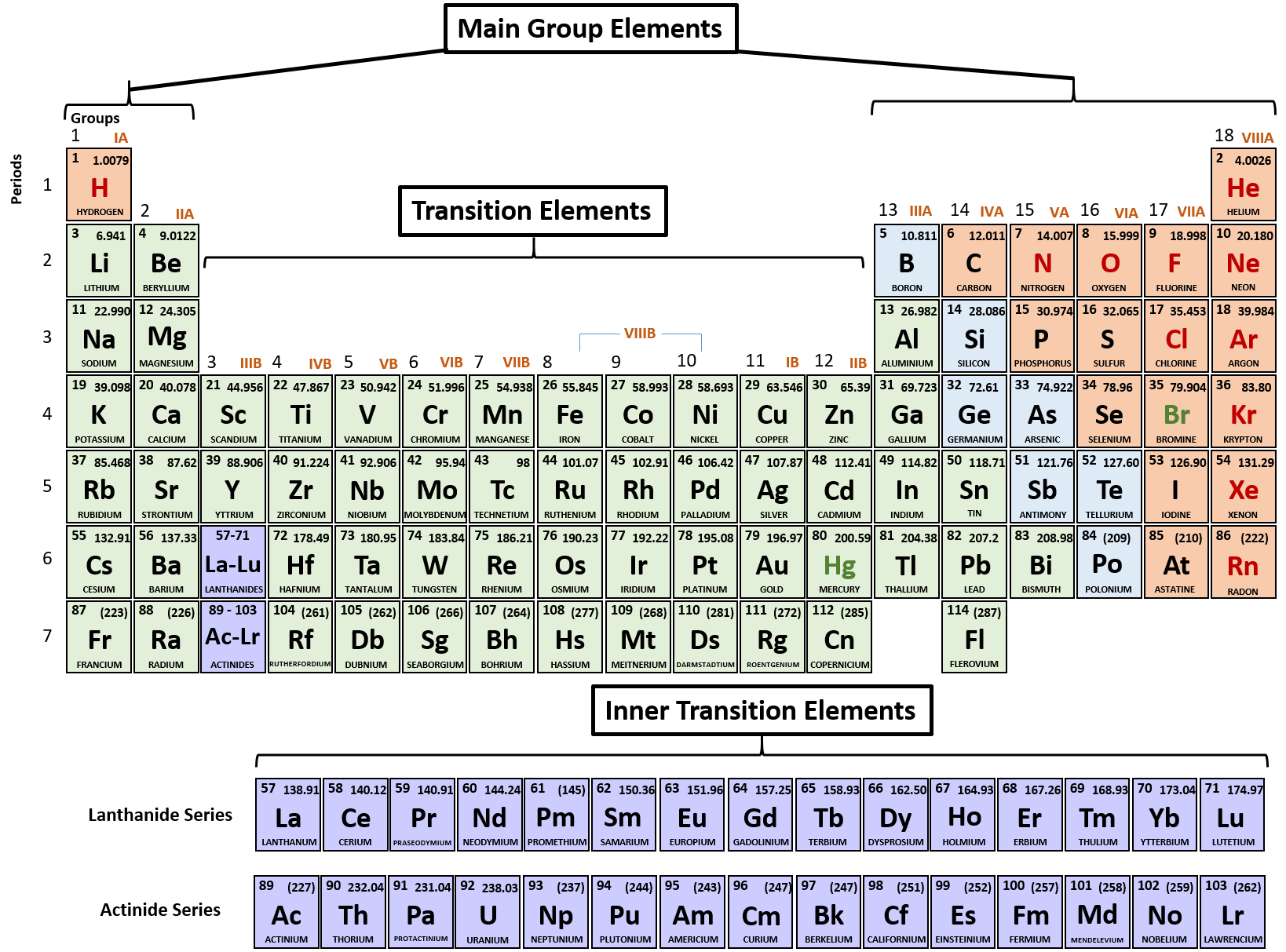
This is because of the basic principle of ‘opposites attract’, where any two objects with opposing electrical charges are attracted towards each other. There is always a force of attraction between two oppositely charged ions. Therefore, the number of negative charges is greater than the number of positive charges and the particle becomes negatively charged overall. If the atom or molecule has gained electrons, the number of electrons is greater than the number of protons. Therefore, the number of positive charges is greater than the number of negative charges and the particle becomes positively charged overall. If the atom or molecule has lost electrons, the number of protons is greater than the number of electrons. Protons are positively charged and electrons are negatively charged. An ion is a charged particle formed when an atom or molecule loses or gains electrons from the outer shells, causing the number of protons and electrons to become unequal.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
